ocean acidity
Ocean acidification is the ongoing decrease in the pH of the Earth's oceans, caused by the uptake of carbon dioxide from the atmosphere, or, simply put, oceans become more acidic as they absorb more carbon from the air.
Carbon in the oceans is present in 3 forms:
- dissolved CO2 (or H2CO3: carbonic acid, a weak acid);
- bicarbonate ions (HCO3); and
- various carbonate ions[1].
When more CO2 is added to the ocean, it causes some carbonic acid to dissociate into bicarbonate, causing the ocean to become more acidic, thus:
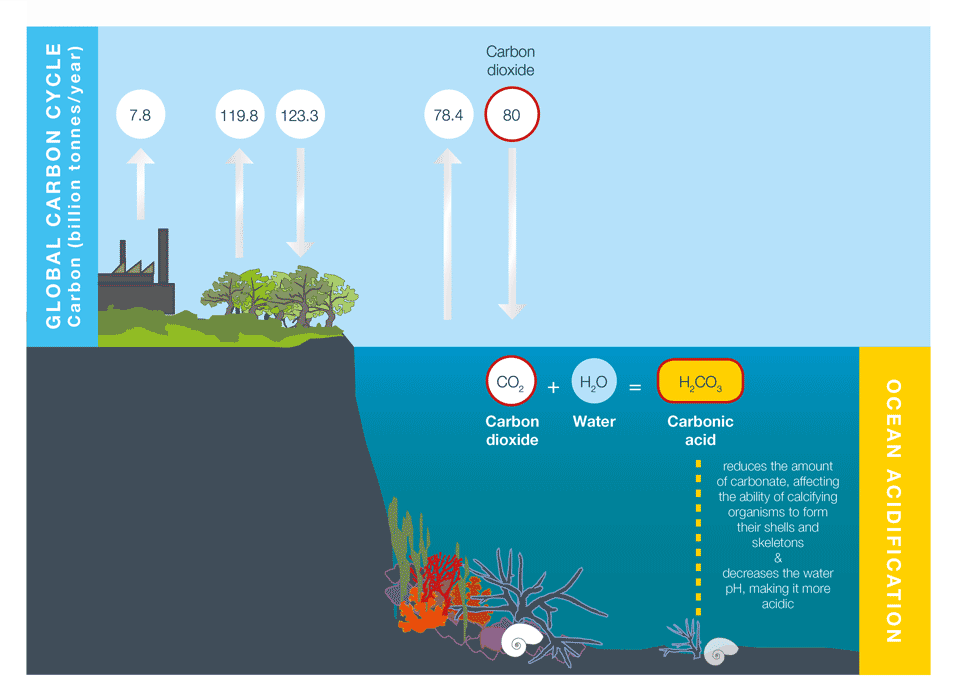 Some of the carbon dioxide emitted to the atmosphere by human activities is absorbed by the oceans. When carbon dioxide combines with water in the ocean it forms carbonic acid, which makes the ocean more acidic and may reduce the ability of calcifying organisms to form their shells and skeletons. Source: Adapted from J. Cook, skepticalscience.com. In turn, adapted from the Coast Adapt site: https://coastadapt.com.au/ocean-acidification-and-its-effects
Some of the carbon dioxide emitted to the atmosphere by human activities is absorbed by the oceans. When carbon dioxide combines with water in the ocean it forms carbonic acid, which makes the ocean more acidic and may reduce the ability of calcifying organisms to form their shells and skeletons. Source: Adapted from J. Cook, skepticalscience.com. In turn, adapted from the Coast Adapt site: https://coastadapt.com.au/ocean-acidification-and-its-effects
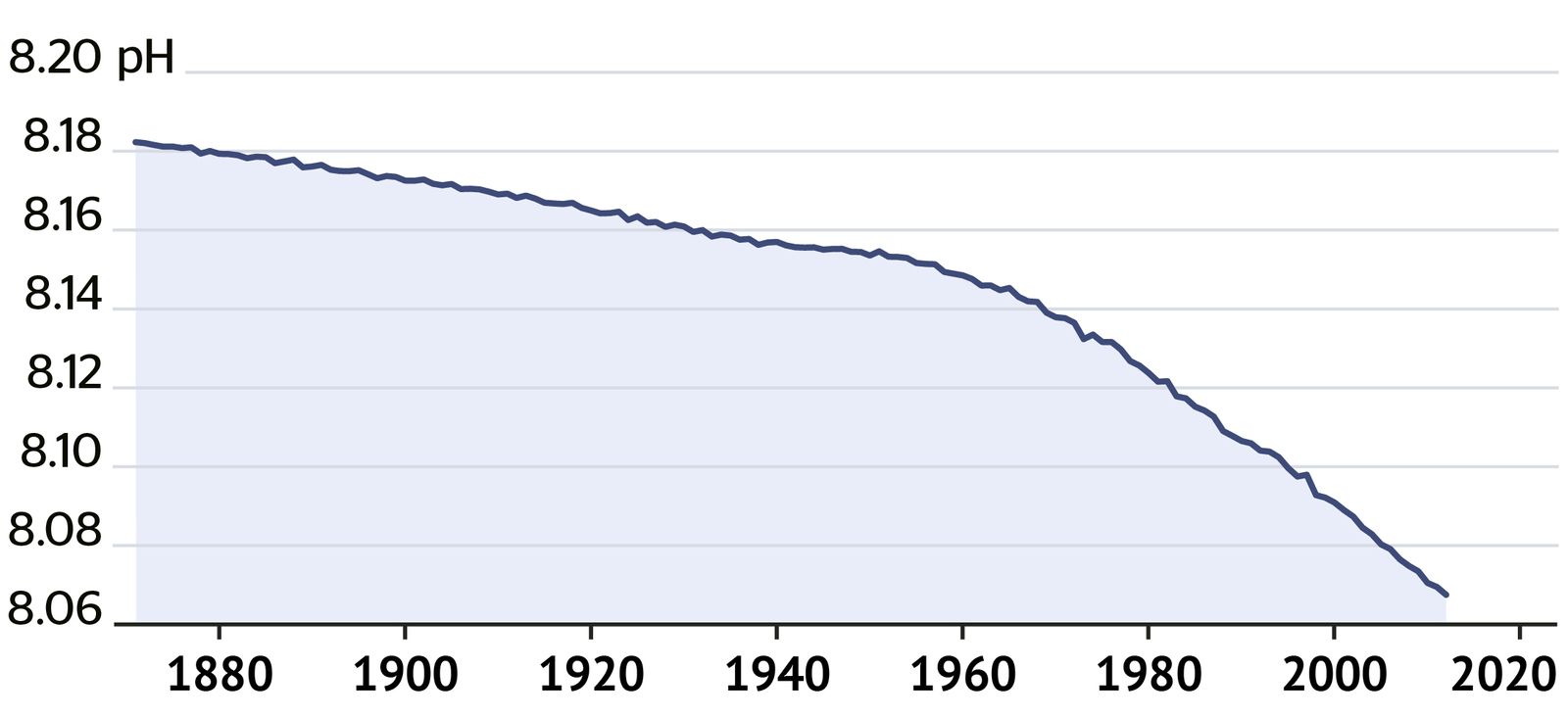 The acidity of waters around Australia is increasing (pH is decreasing).
pH Australian waters. Source: CSIRO.
The acidity of waters around Australia is increasing (pH is decreasing).
pH Australian waters. Source: CSIRO.
Since the beginning of the Industrial Revolution, atmospheric concentration of CO2 has risen from 280 parts per million (ppm) to just over 400 ppm today, an increase of about 30%. That amount would be much higher without oceans which absorb 30 to 40% of the CO2 in the air. As a result, the amount of carbon dioxide absorbed by the upper layer of the oceans is increasing by about 2 billion tons per year. More CO2 in seawater causes chemical reactions which increase acidity, measured as lower pH[2].
Since 1750, oceanic pH has progressed from 8.25 to 8.14 today, The pH scale is a logarithmic scale that usually runs from 1 to 14. Each whole pH value below 7 (the pH of pure water) is ten times more acidic than the higher value and each whole pH value above 7 is ten times less acidic than the one below it. For example, a pH of 3 is ten times more acidic than a pH of 4 and 100 times (10 times 10) more acidic than a pH value of 5. So, a strong acid may have a pH of 1-2, while a strong base may have a pH of 13-14. A pH near 7 is considered to be neutral[3].
Since 1850, the oceans have absorbed between a third and a half of the CO2 humans have released into the atmosphere since, slowing the rate of climate change. During the same period, the acidity of the oceans has increased by 26%, a rate of change roughly 10 times faster than any time in the last 55 million years. Levels are anticipated to be 60%, 100% and 150% more acidic by 2030, 2050 and 2100 respectively. With oceans absorbing about 22 million tonnes of carbon dioxide from the atmosphere a day, seas have already become about 30 per cent more acidic over the past two centuries.
Recent research has revealed that increased acidity can have an impact in undermining important behaviours in marine creatures imperilling their existence[4]. Cold water absorbs more CO2 than warm water does, and more cold water is now open to the air. As the water acidifies, it loses carbonate.
Associated chemical reactions can make it difficult for marine calcifying organisms, such as coral and some plankton, to form shells and skeletons, and existing shells become vulnerable to dissolution. Within the next 15 years, it may no longer contain enough for animals such as sea snails and Alaskan king crabs to construct and maintain their calcium carbonate shells[5]. The rate at which acidification occurs is a determining factor in the extent to which calcifying organisms will be able to adapt.
On current emissions trajectories, global pH levels could drop to as low as 7.8. Even at 8, all sorts of physiological changes can be anticipated, including entire ocean food chains being placed at risk. The impacts of acidification will extend up the food chain to affect economic activities such as fisheries, aquaculture and tourism. Wherever there are marine calcifying organisms, there are risks from ocean acidification.
In 2018-9, a team of scientists exposed four coral and two coralline, or calcifying algal, species to varying ocean acidity levels for a year at the Moorea reef in French Polynesia to test their responses, and found no acclimatisation to the new conditions for over a year, demonstrating that vulnerable reefs may not be able adapt fast enough to cope with climate change.
[1] Associate Professor Michael Box, UNSW (retired), Geoengineering: Messing with the Climate to combat our current mess, WEA course, 16 August 2019.
[2] pH is the negative log of hydrogen ion concentration in a water-based solution. pH is an abbreviation for "power of hydrogen" where "p" is short for the German word for power, potenz and H is the element symbol for hydrogen. The H is capitalized because it is standard to capitalise element symbols. The term "pH" was first described by Danish biochemist Søren Peter Lauritz Sørensen in 1909: https://www.thoughtco.com/what-does-ph-stand-for-608888
[3] Ibid
[4] Danielle L Dixson, “Ocean acidification may alter the behaviours of underwater creatures in disastrous ways”, Scientific American, June 2017, 36-39.
[5] Andy Isaacson, “Into thin ice – the Arctic ice pack is dwindling. What will that do to the planet?” National Geographic, January 2016, 99 at 113; see also the Coast Adapt site: https://coastadapt.com.au/ocean-acidification-and-its-effects
Since 1750, oceanic pH has progressed from 8.25 to 8.14 today, The pH scale is a logarithmic scale that usually runs from 1 to 14. Each whole pH value below 7 (the pH of pure water) is ten times more acidic than the higher value and each whole pH value above 7 is ten times less acidic than the one below it. For example, a pH of 3 is ten times more acidic than a pH of 4 and 100 times (10 times 10) more acidic than a pH value of 5. So, a strong acid may have a pH of 1-2, while a strong base may have a pH of 13-14. A pH near 7 is considered to be neutral[3].
Since 1850, the oceans have absorbed between a third and a half of the CO2 humans have released into the atmosphere since, slowing the rate of climate change. During the same period, the acidity of the oceans has increased by 26%, a rate of change roughly 10 times faster than any time in the last 55 million years. Levels are anticipated to be 60%, 100% and 150% more acidic by 2030, 2050 and 2100 respectively. With oceans absorbing about 22 million tonnes of carbon dioxide from the atmosphere a day, seas have already become about 30 per cent more acidic over the past two centuries.
Recent research has revealed that increased acidity can have an impact in undermining important behaviours in marine creatures imperilling their existence[4]. Cold water absorbs more CO2 than warm water does, and more cold water is now open to the air. As the water acidifies, it loses carbonate.
Associated chemical reactions can make it difficult for marine calcifying organisms, such as coral and some plankton, to form shells and skeletons, and existing shells become vulnerable to dissolution. Within the next 15 years, it may no longer contain enough for animals such as sea snails and Alaskan king crabs to construct and maintain their calcium carbonate shells[5]. The rate at which acidification occurs is a determining factor in the extent to which calcifying organisms will be able to adapt.
On current emissions trajectories, global pH levels could drop to as low as 7.8. Even at 8, all sorts of physiological changes can be anticipated, including entire ocean food chains being placed at risk. The impacts of acidification will extend up the food chain to affect economic activities such as fisheries, aquaculture and tourism. Wherever there are marine calcifying organisms, there are risks from ocean acidification.
In 2018-9, a team of scientists exposed four coral and two coralline, or calcifying algal, species to varying ocean acidity levels for a year at the Moorea reef in French Polynesia to test their responses, and found no acclimatisation to the new conditions for over a year, demonstrating that vulnerable reefs may not be able adapt fast enough to cope with climate change.
[1] Associate Professor Michael Box, UNSW (retired), Geoengineering: Messing with the Climate to combat our current mess, WEA course, 16 August 2019.
[2] pH is the negative log of hydrogen ion concentration in a water-based solution. pH is an abbreviation for "power of hydrogen" where "p" is short for the German word for power, potenz and H is the element symbol for hydrogen. The H is capitalized because it is standard to capitalise element symbols. The term "pH" was first described by Danish biochemist Søren Peter Lauritz Sørensen in 1909: https://www.thoughtco.com/what-does-ph-stand-for-608888
[3] Ibid
[4] Danielle L Dixson, “Ocean acidification may alter the behaviours of underwater creatures in disastrous ways”, Scientific American, June 2017, 36-39.
[5] Andy Isaacson, “Into thin ice – the Arctic ice pack is dwindling. What will that do to the planet?” National Geographic, January 2016, 99 at 113; see also the Coast Adapt site: https://coastadapt.com.au/ocean-acidification-and-its-effects
